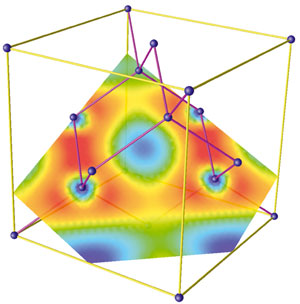
Major challenges lie in unambiguously capturing the atomic-scale fluctuations in water experimentally. Due to the complexity of the H-bond network and its competition with thermal fluctuations, a precise molecular-level understanding of the structure of liquid water remains elusive. This tetrahedral network is realized in the solid phase ice I h, but thermal fluctuations disrupt the H-bond network in the liquid state, with the network fluctuating on picosecond to nanosecond timescales. In an ideal arrangement, water molecules form a tetrahedral network of hydrogen (H) bonds with each vertex being occupied by a water molecule. Despite the apparent simplicity of an H 2O molecule, water in the condensed phase displays a variety of anomalous properties that originate from its complex structure. Without water, life, as we know it, would not exist, earning water the pseudonym “matrix of life,” among others ( 1). 
Water is arguably the most important molecule for life and is involved in almost all biological processes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
